Radiation: Difference between revisions
imported>Anthony.Sebastian (comment of 'rays'.) |
mNo edit summary |
||
| (2 intermediate revisions by one other user not shown) | |||
| Line 1: | Line 1: | ||
{{subpages}} | {{subpages}} | ||
'''Radiation''' is | '''Radiation''' is a flow, through space, of atomic and subatomic particles, such as [[electron]]s (β-radiation) and [[helium]] nuclei (α-radiation), or of high-energy massless particles, [[photon]]s (as in [[gamma radiation|γ-radiation]]). Radiation can also be described as the transmission of ''[[energy]]'' through space, carried by particles, since the moving particles have [[energy]]. Radiation can also be defined as the transport of energy carried by ''[[waves]]'' through space, as one of the important principles of [[quantum mechanics]] states that beams of (sub)atomic particles can be seen as consisting of waves, see [[wave-particle duality]]. | ||
An important form of radiation is [[electromagnetic radiation]], which in the 19th century was seen as a wave phenomenon, but, as was proposed by [[Einstein]] in 1905, can also be seen as consisting of photons—massless particles that propagate through space with the [[speed of light]]. In fact, [[light]] is a form electromagnetic radiation. Electromagnetic radiation covers a large spectrum of wave energy related to wave frequency and amplitude, from low-energy [[radio]] waves to highly energetic [[X-ray]]s and γ-rays. Somewhere in the middle of this spectrum the waves/photons activate receptors in our retinas; we call that radiation visible light. We see the light in many colors. Some animals have retinas sensitive to lower and higher energies; for them infra-red or ultra-violet colors are visible. | An important form of radiation is [[electromagnetic radiation]], which in the 19th century was seen as a wave phenomenon, but, as was proposed by [[Einstein]] in 1905, can also be seen as consisting of photons—massless particles that propagate through space with the [[speed of light]]. In fact, [[light]] is a form electromagnetic radiation. Electromagnetic radiation covers a large spectrum of wave energy related to wave frequency and amplitude, from low-energy [[radio]] waves to highly energetic [[X-ray]]s and γ-rays. Somewhere in the middle of this spectrum the waves/photons activate receptors in our retinas; we call that radiation visible light. We see the light in many colors. Some animals have retinas sensitive to lower and higher energies; for them infra-red or ultra-violet colors are visible. | ||
| Line 6: | Line 6: | ||
{{Image|EMSpec.gif|right|500px|Diagrammatic depiction of the electromagnetic spectrum. Note that as wavelengths become shorter and frequency of wave oscillations increase, the propagated energy increases (expressed in units of electron volts of photon energy). See diagram also in article, [[Electromagnetic radiation]].}} | {{Image|EMSpec.gif|right|500px|Diagrammatic depiction of the electromagnetic spectrum. Note that as wavelengths become shorter and frequency of wave oscillations increase, the propagated energy increases (expressed in units of electron volts of photon energy). See diagram also in article, [[Electromagnetic radiation]].}} | ||
As exemplified above, physicists often use the term 'rays' in referring to electromagnetic radiation, in deference to the straight line transmission of the radiation viewed as particles, such as the light rays from the sun (photons), and when the radiation consists of particles with non-zero rest mass (matter rays). High energy electromagnetic radiation is typically referred to as 'rays' (e.g., γ-rays). | As exemplified above, physicists often use the term 'rays' in referring to electromagnetic radiation, in deference to the straight line transmission of the radiation viewed as particles, such as the light rays from the sun (photons), and when the radiation consists of particles with non-zero rest mass (matter rays). High energy electromagnetic radiation is typically referred to as 'rays' (e.g., X-rays and γ-rays). High-energy X-rays and γ-rays can behave like particles with mass in that they can transfer [[momentum]] (mass x velocity) to electrons they collide with (see [[Compton effect]]). | ||
High-energy radiation, such as X-rays and γ-rays, can ionize matter, i.e., remove electrons from the atoms that constitute matter. For living cells such ionization can be damaging, see [[ionizing radiation]]. | High-energy radiation, such as X-rays and γ-rays, can ionize matter, i.e., remove electrons from the atoms that constitute matter. For living cells such ionization can be damaging, see [[ionizing radiation]]. | ||
| Line 12: | Line 12: | ||
Certain [[atomic nucleus|atomic nuclei]] are spontaneously [[radioactive]], that is, they emit α, β, or γ radiation (or mixtures of the three kinds of radiation). [[Neutrino]]s may also be emitted during nuclear radiation. Highly energetic nuclear radiation can cause injury to living systems. | Certain [[atomic nucleus|atomic nuclei]] are spontaneously [[radioactive]], that is, they emit α, β, or γ radiation (or mixtures of the three kinds of radiation). [[Neutrino]]s may also be emitted during nuclear radiation. Highly energetic nuclear radiation can cause injury to living systems. | ||
As stated, photons travel with the speed of light, which is possible because they have [[rest mass]] zero. The α and β particles (helium nuclei and electrons) have rest mass not equal to zero; the same is true for [[neutrons]] and [[protons]]. According to the theory of [[special relativity]] | As stated, photons travel with the speed of light, which is possible because they have a (theoretical) [[rest mass]] of zero. The α and β particles (helium nuclei and electrons) have rest mass not equal to zero; the same is true for [[neutrons]] and [[protons]]. According to the theory of [[special relativity]] those particles with finite mass cannot achieve the speed of light because at that speed their [[Energy_(science)#Equivalence of energy and mass|relativistic kinetic energy]] would become infinitely large.[[Category:Suggestion Bot Tag]] | ||
Latest revision as of 16:00, 9 October 2024
Radiation is a flow, through space, of atomic and subatomic particles, such as electrons (β-radiation) and helium nuclei (α-radiation), or of high-energy massless particles, photons (as in γ-radiation). Radiation can also be described as the transmission of energy through space, carried by particles, since the moving particles have energy. Radiation can also be defined as the transport of energy carried by waves through space, as one of the important principles of quantum mechanics states that beams of (sub)atomic particles can be seen as consisting of waves, see wave-particle duality.
An important form of radiation is electromagnetic radiation, which in the 19th century was seen as a wave phenomenon, but, as was proposed by Einstein in 1905, can also be seen as consisting of photons—massless particles that propagate through space with the speed of light. In fact, light is a form electromagnetic radiation. Electromagnetic radiation covers a large spectrum of wave energy related to wave frequency and amplitude, from low-energy radio waves to highly energetic X-rays and γ-rays. Somewhere in the middle of this spectrum the waves/photons activate receptors in our retinas; we call that radiation visible light. We see the light in many colors. Some animals have retinas sensitive to lower and higher energies; for them infra-red or ultra-violet colors are visible.
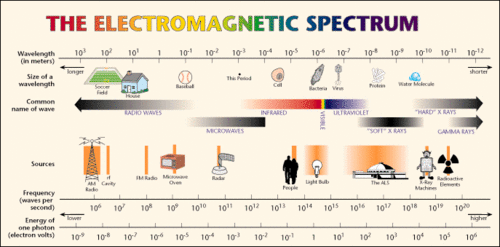
Diagrammatic depiction of the electromagnetic spectrum. Note that as wavelengths become shorter and frequency of wave oscillations increase, the propagated energy increases (expressed in units of electron volts of photon energy). See diagram also in article, Electromagnetic radiation.
As exemplified above, physicists often use the term 'rays' in referring to electromagnetic radiation, in deference to the straight line transmission of the radiation viewed as particles, such as the light rays from the sun (photons), and when the radiation consists of particles with non-zero rest mass (matter rays). High energy electromagnetic radiation is typically referred to as 'rays' (e.g., X-rays and γ-rays). High-energy X-rays and γ-rays can behave like particles with mass in that they can transfer momentum (mass x velocity) to electrons they collide with (see Compton effect).
High-energy radiation, such as X-rays and γ-rays, can ionize matter, i.e., remove electrons from the atoms that constitute matter. For living cells such ionization can be damaging, see ionizing radiation.
Certain atomic nuclei are spontaneously radioactive, that is, they emit α, β, or γ radiation (or mixtures of the three kinds of radiation). Neutrinos may also be emitted during nuclear radiation. Highly energetic nuclear radiation can cause injury to living systems.
As stated, photons travel with the speed of light, which is possible because they have a (theoretical) rest mass of zero. The α and β particles (helium nuclei and electrons) have rest mass not equal to zero; the same is true for neutrons and protons. According to the theory of special relativity those particles with finite mass cannot achieve the speed of light because at that speed their relativistic kinetic energy would become infinitely large.