Van der Waals equation: Difference between revisions
imported>Paul Wormer |
imported>Paul Wormer |
||
| Line 109: | Line 109: | ||
== Other thermodynamic parameters == | == Other thermodynamic parameters == | ||
The extensive volume ''V'' is related to the volume per particle ''v=V/N'' where ''N = nN''<sub>A</sub> is the number of particles in the system. | |||
The equation of state does not give us all the thermodynamic parameters of the system. We can take the equation for the [[Helmholtz energy]] ''A'' | The equation of state does not give us all the thermodynamic parameters of the system. We can take the equation for the [[Helmholtz energy]] ''A'' | ||
| Line 117: | Line 117: | ||
From the equation derived above for ln''Q'', we find | From the equation derived above for ln''Q'', we find | ||
:<math>A(T,V,N)=-NkT\left(1+\ln\left(\frac{(V- | :<math>A(T,V,N)=-NkT\left(1+\ln\left(\frac{(V-nb)T^{3/2}}{N\Phi}\right)\right) | ||
-\frac{ | -\frac{n^2 a}{V}</math> | ||
This equation expresses ''A'' in terms of its [[Thermodynamic potential|natural variables]] ''V'' and ''T'' , and therefore gives us all thermodynamic information about the system. The mechanical equation of state was already derived above | This equation expresses ''A'' in terms of its [[Thermodynamic potential|natural variables]] ''V'' and ''T'' , and therefore gives us all thermodynamic information about the system. The mechanical equation of state was already derived above | ||
:<math>p = -\left(\frac{\partial A}{\partial V}\right)_T | :<math>p = -\left(\frac{\partial A}{\partial V}\right)_T | ||
= \frac{NkT}{V- | = \frac{NkT}{V-nb}-\frac{n^2 a}{V^2}</math> | ||
The entropy equation of state yields the [[entropy]] (''S'' ) | The entropy equation of state yields the [[entropy]] (''S'' ) | ||
:<math>S = -\left(\frac{\partial A}{\partial T}\right)_V | :<math>S = -\left(\frac{\partial A}{\partial T}\right)_V | ||
=Nk\left[ \ln\left(\frac{(V- | =Nk\left[ \ln\left(\frac{(V-nb)T^{3/2}}{N\Phi}\right)+\frac{5}{2} \right]</math> | ||
from which we can calculate the [[internal energy]] | from which we can calculate the [[internal energy]] | ||
:<math>U = A+TS = \frac{3}{2}\,NkT-\frac{ | :<math>U = A+TS = \frac{3}{2}\,NkT-\frac{n^2 a}{V}</math> | ||
Similar equations can be written for the other [[thermodynamic potentials]] and the [[chemical potential]], but expressing any potential as a function of pressure ''p'' will require the solution of a third-order polynomial, which yields a complicated expression. Therefore, expressing the [[enthalpy]] and the [[Gibbs energy]] as functions of their natural variables will be complicated. | Similar equations can be written for the other [[thermodynamic potentials]] and the [[chemical potential]], but expressing any potential as a function of pressure ''p'' will require the solution of a third-order polynomial, which yields a complicated expression. Therefore, expressing the [[enthalpy]] and the [[Gibbs energy]] as functions of their natural variables will be complicated. | ||
Revision as of 07:55, 8 September 2007
In physics, chemistry, and chemical engineering, the van der Waals equation is an equation of state for a fluid composed of particles that have a non-zero size and a pairwise attractive inter-particle force (such as the van der Waals force.) It was derived by Johannes Diderik van der Waals in his doctoral thesis (Leiden 1873), based on a modification of the ideal gas law. The importance of his work was the recognition that the gas and liquid phase of a compound transform continuously into each other. Above the critical temperature there is even no difference between the gas and the liquid phase, which is why it is proper to speak of the equation of a fluid, a generic term for liquid and gas.
Equation
Consider a vessel of volume V filled with n moles of atoms or molecules of a single compound. Let Avogadro's constant be NA, so that the total number of particles in the vessel is nNA, then the van der Waals equation reads
- ,
where
- V is the total volume of the container containing the fluid
- a is a measure of the strength of attraction between the particles
- b is the volume excluded by a mole of particles,
- n is the number of moles
- R is the gas constant, R = NA k, where k is the Boltzmann constant.
The van der Waals parameter b is NA times the volume of a single particle—the volume bounded by the atomic radius. In van der Waals' original derivation, given below, b/NA is four times the volume of the particle. Observe that the pressure p goes to infinity when the container is completely filled with particles so that there is no void space left for the particles to move. This occurs when V = n b.
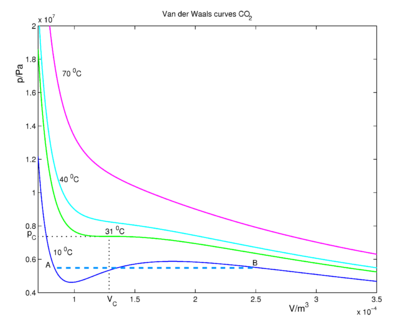
The critical temperature TC is 8a/(27 R b). For constant temperature T = 0.85 TC, and the fairly arbitrary values b = 3 and a = 1, the van der Waals curve has the shape given in the following graph, in which we see pressure versus molar volume Vm.
For large volume the pressure is low and the fluid (which is in the gas phase at low pressures) obeys approximately the ideal gas law p V = R T. If we decrease the volume, the pressure rises until we reach the point C, where condensation (formation of liquid) starts. At this point the van der Waals curve is no longer physical (excluding the possibility of the occurrence of an oversaturated, metastable gas), because from C onward to smaller volume, the pressure stays constant—equal to the vapor pressure of the liquid. All molecules are in the liquid phase at point A, no molecules in the gas phase are left. When we now squeeze the volume at A containing liquid only, the pressure will rise steeply, because the compressibility of a liquid is considerable smaller than that of a gas. Although the wiggles in the van der Waals curve are not physical, the corresponding equation was a great scientific achievement in 1873. Even today it is not possible to give a single equation that describes correctly the phase transition.
Validity
Above the critical temperature the van der Waals equation is an improvement of the ideal gas law, and for lower temperatures the equation is also qualitatively reasonable for the liquid state and the low-pressure gaseous state. However, the van der Waals model cannot be taken seriously in a quantitative sense, it is only useful for qualitative purposes.[1]
In the first-order phase transition range of (p,V,T) (where the liquid phase and the gas phase are in equilibrium) it does not exhibit the empirical fact that p is constant as a function of V for a given temperature.
Derivation
In the usual textbooks one finds two different derivations. One is the conventional derivation that goes back to van der Waals and the other is a statistical mechanics derivation. The latter has its major advantage that it makes explicit the intermolecular potential, which is out of sight in the first derivation.
Conventional derivation
Consider first one mole of gas which is composed of non-interacting point particles that satisfy the ideal gas law
Next assume that all particles are hard spheres of the same finite radius r (the van der Waals radius). The effect of the finite volume of the particles is to decrease the available void space in which the particles are free to move. We must replace V by V-b, where b is called the excluded volume. The corrected equation becomes
The excluded formula is not just equal to the volume occupied by the solid, finite size, particles, but actually four times that volume. To see this we must realize that a particle is surrounded by a sphere of radius d = 2r that is forbidden for the centers of the other particles. If the distance between two particle centers would be smaller than 2r, it would mean that the two particles penetrate each other, which, by definition, hard spheres are unable to do. The excluded volume per particle is , which we must divide by two to avoid overcounting, so that the excluded volume is 4 × 4π r3/3, which is four times the proper volume of the particle. It was a point of concern to van der Waals that the factor four yields actually an upper bound, empirical values for are usually lower. Of course molecules are not infinitely hard, as van der Waals thought, but are often fairly soft.
Next, we introduce a pairwise attractive force between the particles. Van der Waals assumed that, notwithstanding the existence of this force, the density of the fluid is homogeneous. Further he assumed that the range of the attractive force is so small that the great majority of the particles do not feel that the container is of finite size. That is, the bulk of the particles do not notice that they have more attracting particles to their right than to their left when they are relatively close to the left-hand wall of the container. The same statement holds with left and right interchanged. Given the homogeneity of the fluid, the bulk of the particles do not experience a net force pulling them to the right or to the left. This is different for the particles in surface layers directly adjacent to the walls. They feel a net force from the bulk particles pulling them into the container, because this force is not compensated by particles between them and the wall (another assumption here is that there is no interaction between walls and particles). This net force decreases the force exerted onto the wall by the particles in the surface layer. The net force on a surface particle, pulling it into the container, is proportional to the number density . The number of particles in the surface layers is, again by assuming homogeneity, also proportional to the density. In total, the force on the walls is decreased by a factor proportional to the square of the density and the pressure (force per unit surface) is decreased by
- ,
so that
Upon writing n for the number of moles and nVm = V, the equation obtains the form given above,
It is of some historical interest to point out that van der Waals in his Nobel prize lecture gave credit to Laplace for the argument that pressure is reduced proportional to the square of the density.
Statistical thermodynamics derivation
The canonical partition function Q of an ideal gas consisting of N = nNA identical particles, is
where is the thermal de Broglie wavelength,
with the usual definitions: h is Planck's constant, m the mass of a particle, k Boltzmann's constant and T the absolute temperature. In an ideal gas is the partition function of a single particle in a container of volume V. In order to derive the van der Waals equation we assume now that each particle moves independently in an average potential field offered by the other particles. The averaging over the particles is easy because we will assume that the particle density of the van der Waals fluid is homogeneous. The interaction between a pair of particles, which are hard spheres, is taken to be
r is the distance between the centers of the spheres and d is the distance where the hard spheres touch each other (twice the van der Waals radius). The depth of the van der Waals well is .
Because the particles are independent the total partition function still factorizes, , but the intermolecular potential necessitates two modifications to . First, because of the finite size of the particles, not all of V is available, but only , where (just as in the conventional derivation above) . Second, we insert a Boltzmann factor to take care of the average intermolecular potential. We divide here the potential by two because this interaction energy is shared between two particles. Thus
The total attraction felt by a single particle is,
where we assumed that in a shell of thickness dr there are N/V 4π r2dr particles. Performing the integral we get
Hence, we obtain by the use of Stirling's approximation,
It is convenient to take T out of Λ and to rewrite this expression as
where Φ is a constant. From statistical thermodynamics we know that
- ,
so that
Other thermodynamic parameters
The extensive volume V is related to the volume per particle v=V/N where N = nNA is the number of particles in the system.
The equation of state does not give us all the thermodynamic parameters of the system. We can take the equation for the Helmholtz energy A
From the equation derived above for lnQ, we find
This equation expresses A in terms of its natural variables V and T , and therefore gives us all thermodynamic information about the system. The mechanical equation of state was already derived above
The entropy equation of state yields the entropy (S )
from which we can calculate the internal energy
Similar equations can be written for the other thermodynamic potentials and the chemical potential, but expressing any potential as a function of pressure p will require the solution of a third-order polynomial, which yields a complicated expression. Therefore, expressing the enthalpy and the Gibbs energy as functions of their natural variables will be complicated.
Reduced form
Although the material constants a and b in the usual form of the van der Waals equation differs for every single fluid considered, the equation can be recast into an invariant form applicable to all fluids.
Defining the following reduced variables (, is the reduced and critical variables version of , respectively),
- ,
where
The first form of the van der Waals equation of state given above can be recast in the following reduced form:
This equation is invariant (i.e., the same equation of state, viz., above, applies) for all fluids.
Thus, when measured in intervals of the critical values of various quantities, all fluids obey the same equation of state -- the reduced van der Waals equation of state. This is also known as the Principle of corresponding states. In the sense that we have eliminated the appearance of the individual material constants a and b in the equation, this can be considered unity in diversity.
Reference
- ↑ T. L. Hill, Statistical Thermodynamics, Addison-Wesley, Reading (1960), p. 280




















![{\displaystyle \exp[-\phi /(2kT)]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/e5a94595f750dac5aa4133e29923987e04f1e264)




![{\displaystyle \ln Q=N\left(1+\ln {\Bigg [}{\frac {(V-Nb')\,T^{3/2}}{N\Phi }}{\Bigg ]}\right)+{\frac {N^{2}a'}{VkT}},}](https://wikimedia.org/api/rest_v1/media/math/render/svg/55014f0ac9a0dad91981c210af86a007153200cc)





![{\displaystyle S=-\left({\frac {\partial A}{\partial T}}\right)_{V}=Nk\left[\ln \left({\frac {(V-nb)T^{3/2}}{N\Phi }}\right)+{\frac {5}{2}}\right]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/be3fa7b55e9ddcf6a3aecee178267713077872cf)






