Lactam: Difference between revisions
Jump to navigation
Jump to search

imported>David E. Volk mNo edit summary |
imported>David E. Volk (picture of lactam structurea) |
||
| Line 1: | Line 1: | ||
{{subpages}} | {{subpages}} | ||
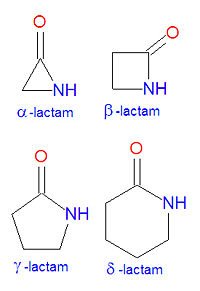
In chemistry, a '''lactam''' is a cyclic [[amide]]. The name is derived from two chemical terms, [[lactone]], referring to a cyclic ketone, and [[amide]], a compound containing a [[nitrogen]] atom next to a [[carbonyl]] group. Lactams are named according to the size of the cyclic ring in the lactam: <math>\alpha</math>-lactams, <math>\beta</math>-lactams, <math>\gamma</math>-lactams and <math>\delta</math>-lactams contain rings made of three, four, five | [[Image:Lactams.jpg|right|thumb|200px|Core lactam structures.]] | ||
In chemistry, a '''lactam''' is a cyclic [[amide]]. The name is derived from two chemical terms, [[lactone]], referring to a cyclic ketone, and [[amide]], a compound containing a [[nitrogen]] atom next to a [[carbonyl]] group. Lactams are named according to the size of the cyclic ring in the lactam: <math>\alpha</math>-lactams, <math>\beta</math>-lactams, <math>\gamma</math>-lactams and <math>\delta</math>-lactams contain rings made of three, four, five or six atoms, respectively. <math>\alpha</math>-lactams are also called aziridinones. Many widely used antibiotic drugs, including the[[penicillin]]s and [[cephalosporin]]s, owe their activity to the presence of a <math>\beta</math>-lactam structure. The lactams may have substitutions added to the nitrogen atom or any of the non-carbonyl carbon atoms in the base structure. | |||
Revision as of 13:34, 17 May 2008
In chemistry, a lactam is a cyclic amide. The name is derived from two chemical terms, lactone, referring to a cyclic ketone, and amide, a compound containing a nitrogen atom next to a carbonyl group. Lactams are named according to the size of the cyclic ring in the lactam: -lactams, -lactams, -lactams and -lactams contain rings made of three, four, five or six atoms, respectively. -lactams are also called aziridinones. Many widely used antibiotic drugs, including thepenicillins and cephalosporins, owe their activity to the presence of a -lactam structure. The lactams may have substitutions added to the nitrogen atom or any of the non-carbonyl carbon atoms in the base structure.